Other grades of this product :
| Tetrachlorvinphos Basic information |
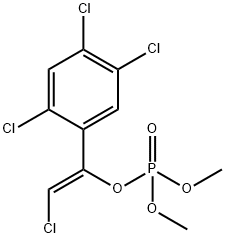
| Product Name: | Tetrachlorvinphos | | Synonyms: | ncic00168;o,o-dimethyl-o-2-chlor-1-(2,4,5-trichlorphenyl)-vinyl-phosphat;Phosphoric acid, 2-chloro-1-(2,4,5-trichlorophenyl)ethenyl dimethyl ester, (Z)-;phosphoricacid,2-chloro-1-(2,4,5-trichlorophenyl)ethenyldimethylester;phosphoricacid,2-chloro-1-(2,4,5-trichlorophenyl)vinyldimethylester;Rabon;Rabond;ROL | | CAS: | 22248-79-9 | | MF: | C10H9Cl4O4P | | MW: | 365.96 | | EINECS: | 244-865-4 | | Product Categories: | | Mol File: | 22248-79-9.mol |
| Tetrachlorvinphos Chemical Properties |
| Tetrachlorvinphos Usage And Synthesis |
| Description | Tetrachlorvinphos was initially registered for use in the United
States in 1966 for use on various food crops, livestock, and pet
animals, and in around buildings. Its use on food crops were
voluntarily canceled in the United States in 1987; however, it is
used on food crops in developing countries. Tetrachlorvinphos
is sold under the trade names Rabon and Gardona. | | Chemical Properties | Powder.Partially soluble in
chloroform; slightly soluble in water. | | Chemical Properties | Technical tetrachlorvinphos is a tan-to-brown crystalline
solid. Tetrachlorvinphos is stable at ,100 C and slowly
hydrolyzed at 50°C. Aromatic odor.
Soluble in water at 24°C 15 ppm; limited
solubility in most aromatic hydrocarbons. | | Uses | Insecticide. | | Uses | Tetrachlorvinphos is used to control lepidopterous and dipterous
larvae in fruit and lepidopterous larvae in cotton, maize, rice, tobacco and
vegetables. It is also used against nuisance flies in animal houses, animal
ectoparasites and stored product pests. | | Uses | Tetrachlorvinphos is commonly used as a feed additive to
control flies in livestock and as dusts, sprays, dips, and collar
ingredient to control ticks and fleas on domestic pets. It is
extensively used in poultry. In horses, its formulations are
commonly used as a feed additive (feed-through tetrachlorvinphos)
larvicide. In addition, tetrachlorvinphos is also
used in the control of public health pests, manure flies associated
with livestock, and poultry as a feed additive. | | Hazard | Cholinesterase inhibitor. Questionable carcinogen. | | Carcinogenicity | When rats were given diets with
0, 4250, or 8500 ppm tetrachlorvinphos for 80 weeks,
both males and females had a high incidence of thyroid
C-cell hyperplasia, and females had increased incidences of
adrenal cortical adenomas and thyroid C-cell adenomas
. | | Environmental Fate | Tetrachlorvinphos is nonpersistent in the environment. The
primary route of dissipation is through biotic degradation.
Based on its use pattern, risks of contamination of groundwater
or surface water by tetrachlorvinphos are minimal. | | Metabolic pathway | The chemical structure of tetrachlorvinphos is very close to that of
chlorfenvinphos and the routes of metabolic breakdown have been
shown to be very similar. Technical tetrachlorvinphos is usually >95%
Z-isomer, unlike chlorfenvinphos which is an E/Z mixture. As with
chlorfenvinphos, the major routes of detoxification are by dealkylation
and hydrolysis to yield desmethyltetrachlorvinphos and 2,2’,4’,5’-
tetrachloroacetophenone plus dimethyl phosphate, respectively. Further
metabolism of the chloroacetophenone moiety then leads, via reduction
or hydrolysis and glutathione-dependent displacement of the
side chain chlorine substituent, to the formation of 1-(2,4,5-trichlophenyl)
ethane-l,Z-diol and 1-(2,4,5-trichlorophenyl)ethan-l-owl hich are
conjugated with glucose or glucuronic acid to afford the ultimate
metabolites. Oxidation of the β carbon atom to give 2,4,5-trichloromandelic
acid followed by decarboxylation leads to the formation
of 2,4,5-trichlorobenzoic acid which is conjugated with glycine in some
mammals as the final metabolite. The metabolic routes were summarised
by Beynon et al. (1973). | | Degradation | Tetrachlorvinphos is hydrolysed slowly in neutral, acidic and slightly
alkaline aqueous solutions but hydrolysed rapidly in strongly alkaline
solutions to metabolites 3 and 5 (PM). Dureja et al. (1987) reported
the photochemical degradation of tetrachlorvinphos in water, ethanol,
ether and hexane irradiated with a xenon lamp. In polar solvents, the
main product was desmethyltetrachlorvinphos (2), whereas in non-polar
solvents such as hexane the reaction yielded dimethyl phosphate (3),
2,4,5-trichloroacet ophenone (4) and 2,2’,4’,5’-tetrachloroacetophenone
(5). Interconversion of the Z- and Ε-isomers has been observed on
leaves (Beynon and Wright, 1969). These pathways are shown in
Scheme 1. | | Toxicity evaluation | Acute oral LD50 for rats: 4,000-5,000 mg/kg |
| Tetrachlorvinphos Preparation Products And Raw materials |
|