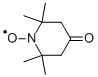
| 4-Oxo-2,2,6,6-tetramethylpiperidinooxy Basic information |
| Product Name: | 4-Oxo-2,2,6,6-tetramethylpiperidinooxy |
| Synonyms: | 4-Oxo-2,2,6,6-tetramethyl-1-piperidinyloxy, free radical;4-OXO-TEMPO, FREE RADICAL 80+%;4-OXO-TEMPO, FREE RADICAL 99+%;4-OXO-2,2,6,6-TETRAMETHYLPIPERIDINOXY;4-Oxo-2,2,6,6-tetramethylpiperidine-1-oxyl(4-Oxo-TEMPO,freeradical);4-Oxo-2,2,6,6;4-Oxo-2,2,6,6-trtramethyl-1-piperidi nyloxy,free radical;2,2,6,6-Tetramethyl-4-piperidone 1-oxyl, free radical |
| CAS: | 2896-70-0 |
| MF: | C9H16NO2* |
| MW: | 170.23 |
| EINECS: | 220-778-7 |
| Product Categories: | Industrial/Fine Chemicals;Spin Labeling Compounds;Analytical Chemistry;ESR Spectrometry;Spin Labels |
| Mol File: | 2896-70-0.mol |
| 4-Oxo-2,2,6,6-tetramethylpiperidinooxy Chemical Properties |
| Melting point | 33-38 °C |
| Boiling point | 299.88°C (rough estimate) |
| density | 1.0710 (rough estimate) |
| vapor pressure | 2.254Pa at 25℃ |
| refractive index | 1.4640 (estimate) |
| storage temp. | 2-8°C |
| form | Needles, Crystals or Crystalline Powder |
| color | Orange |
| Water Solubility | Soluble in water (partly), acetone, DMSO, 100% ethanol, and diethylether. Insoluble in water. |
| BRN | 1818579 |
| LogP | 0.28 at 22℃ |
| CAS DataBase Reference | 2896-70-0(CAS DataBase Reference) |
| NIST Chemistry Reference | 1-Piperidinyloxy, 2,2,6,6-tetramethyl-4-oxo-(2896-70-0) |
| EPA Substance Registry System | 1-Piperidinyloxy, 2,2,6,6-tetramethyl-4-oxo- (2896-70-0) |
| Safety Information |
| Risk Statements | 36/37/38 |
| Safety Statements | 24/25 |
| RIDADR | UN 3077 9 / PGIII |
| WGK Germany | 3 |
| RTECS | TN9200000 |
| TSCA | Yes |
| HS Code | 29333990 |
| MSDS Information |
| Provider | Language |
|---|---|
| 4-Oxo-TEMPO | English |
| ACROS | English |
| SigmaAldrich | English |
| ALFA | English |
| 4-Oxo-2,2,6,6-tetramethylpiperidinooxy Usage And Synthesis |
| Description | 2,2,6,6-Tetramethylpiperidin-1-oxyl (TEMPO) is a stable radical that will react with reactive oxygen species (ROS). This conversion, followed by ESR, provides an indirect way to monitor ROS production in biological systems. TEMPONE is the 4-oxo derivative of TEMPO. In addition to possible uses as a spin trap, this nitroxyl radical can be used in hydrogen transfer experiments and as a polarizing agent in dynamic nuclear polarization NMR spectroscopy. TEMPONE and other nitroxyl radicals have also been shown to reduce mean arterial pressure in spontaneously hypertensive rats when administered intravenously. |
| Chemical Properties | Pink Needles |
| Uses | A spin label for ESR studies |
| Uses | Free-radical nitroxide spin probe typically used for:
|
| General Description | 4-Oxo-TEMPO is formed by the reaction of 2,2,6,6-tetramethyl-4-piperidone (4-oxo-TMP) and 1O2. It is a stable paramagnetic product formed during the irradiation of TiO2 in aqueous suspensions. |
| Flammability and Explosibility | Nonflammable |
| 4-Oxo-2,2,6,6-tetramethylpiperidinooxy Preparation Products And Raw materials |
Please leave a message for us or use the following ways to contact us, we will reply to you as soon as possible, and provide you with the most sincere service, thank you.