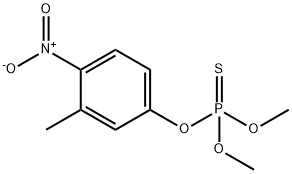
Other grades of this product :
| Fenitrothion Chemical Properties |
| Melting point | 3.4°C | | Boiling point | 140-145°C (0.05 torr) | | density | 1.3227 | | vapor pressure | 1.5 x 10-2 Pa (20 °C) | | refractive index | nD25 1.5528 | | Fp | >100 °C | | storage temp. | APPROX 4°C
| | form | neat | | Specific Gravity | 1.328 (20℃) | | Water Solubility | 0.003 g/100 mL | | Merck | 13,4003 | | BRN | 8983553 | | LogP | 3.319 at 25℃ | | CAS DataBase Reference | 122-14-5(CAS DataBase Reference) | | NIST Chemistry Reference | Fenitrothion(122-14-5) | | EPA Substance Registry System | Fenitrothion (122-14-5) |
| Fenitrothion Usage And Synthesis |
| Description | Fenitrothion is a selective acaricide and contact insecticide of low ovicidal properties. It is a brown to yellow liquid with a characteristic odour. Fenitrothion belongs to the organophosphate family of insecticides. It acts by inhibiting cholinesterase, thus it is an effective pesticide against a wide range of pests such as chewing, penetrating, and sucking insect pests. These pests include locusts, coffee leaf miners, wheat bugs, rice stem borers, grain weevils, and grain beetles on orchard fruits, cereals, cotton, forests, and vegetables. Fenitrothion is also an effective household insecticide as well as vector control agent for malaria.
| | History | Both Bayer Leverkusen and Sumitomo Chemical Company introduced Fenitrothion in 1959. Fenitrothion has minimal toxicity as compared to parathion, albeit with a range of insecticidal activity that is very similar. Fenitrothion is marketed in emulsifiable concentrate, dust, fogging concentrate, flowable, wettable powder formulations, and oil-based liquid spray.
| | Toxicity Effects | Acute Toxicity
Fenitrothion has acute toxicity to mammals, which is normally considered to be low. However, tests done on rats with doses considerably higher than those of applied for parathion reveal typical symptoms of acute poisoning. It also reduced the energy of birds in acute doses. Acute toxicity reported for a human female was a TDIo of 800 mg/kg.
Chronic Toxicity
In humans, chronic symptoms include fatigue, general malaise, headache, anorexia, and loss of memory, thirst, cramps, loss of weight, tremors, and muscular weakness. Half of the fenitrothion minimally effective dose changed the thyroid structure of a freshwater murrel.
| | Chemical Properties | Clear Yellow Oil | | Chemical Properties | Pure fenitrothion is a yellowish brown liquid with an unpleasant odor. It is insoluble in water, but readily soluble in common organic solvents, such as acetone, alcohol, benzene, chlorinated hydrocarbons, dichloromethane, 2-propanol, toluene, in ethers, methanol, and xylene. It decomposes explosively. Fenitrothion is a contact insecticide and a selective acaricide of low ovicidal properties. Fenitrothion is effective against a wide range of pests, namely, penetrating, chewing, and sucking insect pests (coffee leaf-miners, locusts, rice stem borers, wheat bugs, fl our beetles, grain beetles, grain weevils) on cereals, cotton, orchard fruits, rice, vegetables, and forests. It may also be used as a fl y, mosquito, and cockroach residual contact spray for farms and public health programs. Fenitrothion is also effective against household insects and all nuisance insects. WHO confi rmed its effectiveness as a vector control agent for malaria. It is extensively used in other countries, including Japan, where parathion has been banned. Occupational workers are exposed to fenitrothion during mixing, loading/transportation, and fi eld applications. | | Chemical Properties | Fenitrothion is a volatile brownish-yellow
oil. | | Uses | Insecticide. | | Uses | Fenitrothion is used to control sucking, chewing and boring
insects in cereals, soft fruit, tropical fruit, vines, sugar cane, vegetables,
turf and forestry. It is also used as a public health insecticide for the
control of flies, cockroaches and mosquitoes. Other uses are for the control
of stored product pests and locusts. | | Definition | ChEBI: An organic thiophosphate that is O,O-dimethyl O-phenyl phosphorothioate substituted by a methyl group at position 3 and a nitro group at position 4. | | General Description | Brownish-yellow oil. Used as a selective acaricide and a contact and stomach insecticide against chewing and sucking insects on rice, orchard fruits, vegetables, cereals, cotton and forest. Also used against flies, mosquitoes, and cockroaches. | | Reactivity Profile | Organophosphates, such as Fenitrothion, are susceptible to formation of highly toxic and flammable phosphine gas in the presence of strong reducing agents such as hydrides. Partial oxidation by oxidizing agents may result in the release of toxic phosphorus oxides. | | Hazard | Cholinesterase inhibitor, use may be
restricted. | | Health Hazard | Fenitrothion is toxic to animals and humans. After prolonged periods of exposures to high concentrations of fenitrothion, occupational workers show poisoning. The symptoms include, but are not limited to, general malaise, fatigue, headache, loss of memory and ability to concentrate, anorexia, nausea, thirst, loss of weight, cramps, muscular weakness, and tremors, and at suffi ciently high dosage produce typical cholinergic poisoning. The formulation product, sumithion 50EC, causes delayed neurotoxicity in adult rats, as well as humans. | | Health Hazard | Fenitrothion is an organophosphate insecticide. It is a highly toxic cholinesterase inhibitor, that acts on the nervous system. Does not cause delayed neurotoxicity and contact produces little irritation. | | Fire Hazard | When heated to decomposition, Fenitrothion emits very toxic fumes of oxides of nitrogen, phosphorus and sulfur. Decomposition at 212-284F produces a mixture of organophosphorus polymers. Unstable in alkaline media. Stable for 2 years if stored at 68-77F. Do not store above 104F. | | Flammability and Explosibility | Notclassified | | Agricultural Uses | Insecticide, Acaracide: Not approved for use in EU countries. Registered
for use in the U.S. This is a selective acaricide and a contact
and stomach insecticide. Fenitrothion is a contact insecticide
and selective acaricide of low ovicidal properties. It is
considered an acetylcholinesterase inhibitor. Fenitrothion
is effective against a wide range of pests, i.e. penetrating,
chewing and sucking insect pests (coffee leafminers,
locusts, rice stem borers, wheat bugs, flour beetles, grain
beetles, grain weevils) on cereals, cotton, orchard fruits,
rice, vegetables, and forests. It may also be used as a fly,
mosquito, and cockroach residual contact spray for farms
and public health programs. Fenitrothion is also effective
against household insects and all of the nuisance insects
listed by the World Health Organization. Its effectiveness as
a vector control agent for malaria is confirmed by the World
Health Organization. Fenitrothion is non-systemic, and
non-persistent. Fenitrothion was introduced in 1959 by both
Sumitomo Chemical Company and Bayer Leverkusen and
later by American Cyanamid Company. Fenitrothion is far
less toxic than parathion with a range of insecticidal activity
that is very similar and is similar enough in structure to be
produced in the same factories. The difference in precursor
chemicals might make it somewhat more expensive, but it is
heavily used in other countries, including Japan, where parathion
has been banned. Fenitrothion comes in dust, emulsifiable
concentrate, flowable, fogging concentrate, granules,
ULV, oil-based liquid spray, and wettable powder formultaions.
It is compatible with other neutral insecticides. | | Trade name | ACCOTHION®; ACEOTHION®; AGRIA
1050®; AGRIYA 1050®; AGROTHION®; AMERICAN
CYANAMID CL-47,300®; ARBOGAL®; BAY 41831®;
BAYER 41831®; BAYER S 5660®; CEKUTROTHION®;
CL 47300®; CP47114®; CYFEN®; CYTEL®;
CYTEN®; DICATHION®; DICOFEN®; DYBAR®;
EI 47300®; FALITHION®; FENITEX®; FENITOX®;
FENSTAN®; FOLETHION®; FOLITHION®; H-35-F
87 (BVM)®; 8057HC®; KALEIT®; KEEN SUPERKILL
ANT AND ROACH EXTERMINATOR®; KILLGERM
TETRACIDE INSECTICIDAL SPRAY®; KOTION®;
MEP (PESTICIDE)®; METATHION®; METATHIONE®;
METATION®; MICROMITE®; MONSANTO CP
47114®; NITROPHOS®; NOVATHION®; NUVAND®;
NUVANOL®; OLEOSUMIFENE®; OMS 43®;
OVADOFOS®; PENNWALT C-4852; PESTROY®; S
112A®; S 5660®; SMT®; SUMITHION®[C]; TURBAIR
GRAIN STORAGE INSECTICIDE®; VERTHION® | | Safety Profile | Poison by ingestion,
inhalation, intravenous, and intraperitoneal
routes. Moderately toxic by skin contact,
intratracheal, and subcutaneous routes.
Human systemic effects: coma, diarrhea,
dyspnea, gastrointestinal changes,
hypermodtty, nausea or vomiting,
respiratory depression. Mutation data
reported. When heated to decomposition it
emits very toxic fumes of NOx, POx, and
sox | | Potential Exposure | A potential danger to those involved
in the manufacture, formulation, and application of this
insecticide. It is a selective acaricide; and a contact and
stomach insecticide. Used to control chewing and sucking
insects on rice, orchard fruit; vegetables, cereals, cotton,
and in forests. Also protects against flies, mosquitoes, and
cockroaches | | Metabolic pathway | Fenitrothion is a non-systemic insecticide, the biotransformations and
environmental fate of which have been intensively studied and reviewed.
Metabolism has been studied in mammals (including humans), birds,
fish, crustacea, molluscs, insects, algae, plants and soil. The major routes
of biotransformation involve desulfuration to the oxon analogue
(fenitrooxon) and hydrolysis to give dimethyl phosphate, O,O-dimethyl
phosphorothioate and 3-methyl-4-nitrophenol. Demethylation to give
desmethylfenitrothion and its decomposition products, reduction of
the nitro group and oxidation of the ring methyl group also
occur. Demethylation via glutathione-S-methyl transferases in the liver
is a particularly important mechanism in mammals. Reduction of
the nitro group to an amino group is important in anaerobic soils
and ruminants but this has also been shown to occur in rats, rabbits
and humans where it is presumably carried out by bacteria in the gut.
Oxidation of the 3-methyl group to hydroxymethyl and carboxylate
has been shown to be a degradative route in birds. The major routes of
phase II metabolism involve conjugation of 3-methyl-4-nitrophenol to the
glucoside in plants and insects, to the sulfate ester in birds and the sulfate
ester and glucuronide in mammals. | | Metabolism | The main biotransformation routes involve oxidative
desulfuration to the oxon and dearylation to give
dimethyl hydrogen phosphate, O,O-dimethyl hydrogen
phosphorothioate and 3-methyl-4-nitrophenol. Demethylation
dependent on glutathion-S-alkyl transferase is particularly
important in mammals.Oxidation of the 3-methyl
group to hydroxymethyl and then carboxyl group is also
a degradative route. Reduction of the nitro group to an
amino group occurs in anaerobic soils and ruminants. The
DT50 in soils under upland and submerged conditions were
12–28 and 4–20 d, respectively. | | Shipping | UN3017 Organophosphorus pesticides, liquid,
toxic, flammable, flash point not ,23C, Hazard class: 6.1;
Labels: 6.1-Poisonous materials, 3-Flammable liquid.
UN2810 Toxic liquids, organic, n.o.s., Hazard Class: 6.1;
Labels: 6.1-Poisonous materials, Technical Name Required | | Toxicity evaluation | The acute oral LD50 values in mammals
range from 330 mg/kg in rats to 1850 mg/kg in the guineapig.
Inhalation LC50 (4 h) for rats is >1.2 mg/L air. NOEL
(2 y) for rats and mice is 10 mg/kg diet (0.5 mg/kg/d). ADI
is 5 μg/kg b.w. | | Degradation | Fenitrothion is relatively stable to hydrolysis under normal conditions
(PM). Mikami et al. (1985) reported the hydrolysis of fenitrothion between
pH 5 and 10. Below pH 7 hydrolysis occurred by a pH-independent
mechanism and above pH 9 by a base-catalysed process. At intermediate
pH values both mechanisms were operative. The major mechanism below
pH 8 was demethylation to give desmethylfenitrothion and above
pH 9, cleavage of the P-O-aryl bond gave 3-methyl-4-nitrophenol. | | Incompatibilities | Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases,strong acids, oxoacids, epoxides. Strong oxidizers may cause
release of toxic phosphorus oxides. Organophosphates, in the
presence of strong reducing agents such as hydrides, may
form highly toxic and flammable phosphine gas. Keep away
from alkaline materials | | Waste Disposal | Incineration (for large
amounts); alkaline hydrolysis and landfill (for small
amounts). In accordance with 40CFR165, follow recommendations for the disposal of pesticides and pesticide containers. Must be disposed properly by following package label
directions or by contacting your local or federal environmental
control agency, or by contacting your regional EPA office. |
| Fenitrothion Preparation Products And Raw materials |
|